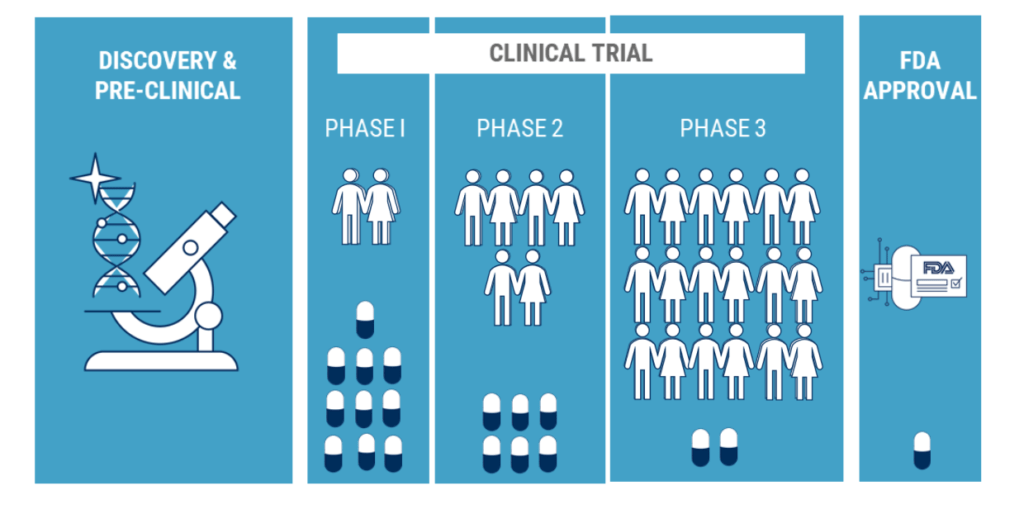
Clinical trials tend to have a cloud of negative talk hovering over them. Participating in something that involves your health can be daunting and scary, and unfortunately, this fear has led to some misconceptions about participating in clinical research trials.
Here are some of the common Myths/Facts surrounding clinical trial participation:
Myth: Clinical trials are dangerous.
Fact: There will always be risks associated with medical research when testing new and unproven treatment options. Uncomfortable side effects may occur. However, clinical trials are federally regulated and there are ethical and legal codes that apply to each study. If you are considering participating it is important to discuss risks and benefits with your doctor. All studies are voluntary and participants can withdraw at any time.
Myth: The quality of care is not as good as the care I currently receive with my doctor.
Fact: Clinical trial participants receive a high level of care and are highly monitored for any side effects that may occur. All participants are able to discuss any areas of concern with the study staff, and the participants primary care provider may also be involved. For some living with Alzheimer’s disease, research shows that they do better while participating in clinical trials.
Myth: Clinical trials will not accept me as a participant because I have another disease or condition.
Fact: Other chronic medical conditions may be present along with Alzheimer’s disease, and some clinical trials may be designed to accommodate this. Each study has its own inclusion and exclusion criteria. When discussing participation during the informed consent process, be sure to let the study team know of any medical conditions that are present.
Myth: If clinical trials are so important, my doctor will encourage me to participate.
Fact: There are hundreds of clinical research studies being done across the country. Your physician may not be aware of which of these studies are in your area, or which studies you may be eligible to participate in. Be sure to discuss any clinical trial opportunities you find with your doctor.
Myth: Clinical trials are not confidential.
Fact: When you enroll in a study there are pieces of personal information that will need to be collected and shared with the study team and study sponsor. This generally includes date of birth, medical history, any results collected throughout the study etc. This information is kept confidential to an extent which means that it is given a blinded number to protect your identity throughout the study. Although your information is shared with the study sponsor and FDA, it is our highest priority to maintain our participants information and dignity.
Myth: The study site/team knows who is actually getting the drug.
Fact: In randomized clinical trials there is a group receiving a placebo and a group receiving the study drug. Neither participants nor study team is aware of which participant is receiving a placebo or not. This information is not released until the closing of each study. Make sure that you are okay with the potential chance of getting a placebo when considering participation in a clinical trial.
Myth: The drugs being tested in clinical trials haven’t been tested on humans yet, I’d just be a “guinea pig.”
Fact: The drugs tested in clinical trials have been researched and evaluated multiple times before they can be used in a clinical trial.
Myth: Clinical Trials don’t work.
Fact: Clinical trials are needed to find safe and effective new treatments for various medical conditions. Some trials show more success than others, but not all trials are failures.
Here are two videos that may be helpful when considering a clinical trial. Clinical Trials.gov also has a helpful page with Questions To Ask, and the Alzheimer’s Association has a page discussing How Clinical Trials Work.
Sources:
10 Misconceptions About Clinical Trials Debunked [Internet]. Rox Medical. 2018 [cited 2019Apr29]. Available from: https://www.roxmedical.com/news/10-clinical-trial-misconceptions/
Clinical Trials: Myths vs. Facts [Internet]. Alzheimer’s Disease and Dementia. [cited 2019Apr29]. Available from: https://www.alz.org/alzheimers-dementia/research_progress/clinical-trials/myths-vs-facts
How Clinical Trials Work [Internet]. Alzheimer’s Disease and Dementia. [cited 2019Apr29]. Available from: https://www.alz.org/alzheimers-dementia/research_progress/clinical-trials/how-trials-work
Learn About Clinical Studies [Internet]. ClinicalTrials.gov. [cited 2019Apr29]. Available from: https://clinicaltrials.gov/ct2/about-studies/learn
